“In this first-of-its-kind human observational report, off-label use of Ivermectin+Mebendazole showed a remarkable 84% Clinical Benefit Ratio in the treatment of cancer. These results indicate that the inexpensive and safe off-label applications of these medications could be an important complement in the treatment of cancer,” – Dr. Peter McCullough, Chief Scientific Officer for The Wellness Company
(Boca Raton, FL) – Doctors from The Wellness Company announced this week that they had completed a first-of-its-kind human observational report of the application of Ivermectin+Mebendazole in the treatment of cancer. The manuscript is currently under peer review.
Dr. Peter McCullough, Chief Scientific Officer for The Wellness Company, and one of the authors of the manuscript, praised the results of this groundbreaking analysis. “In this first of its kind human observational analysis, off-label use of Ivermectin+Mebendazole showed a remarkable 84% Clinical Benefit Ratio in the treatment of cancer. These results indicate that the inexpensive and safe off-label applications of these medications could be an important breakthrough in the treatment of cancer.”
In addition to Dr. Peter McCullough, the manuscript was authored by Nicolas Hulscher, MPH, Kelly Victory, MD, Drew Pinsky, MD, James Thorp, MD, Peter Gillooly, MSc, and Harvey Risch, MD, PhD.
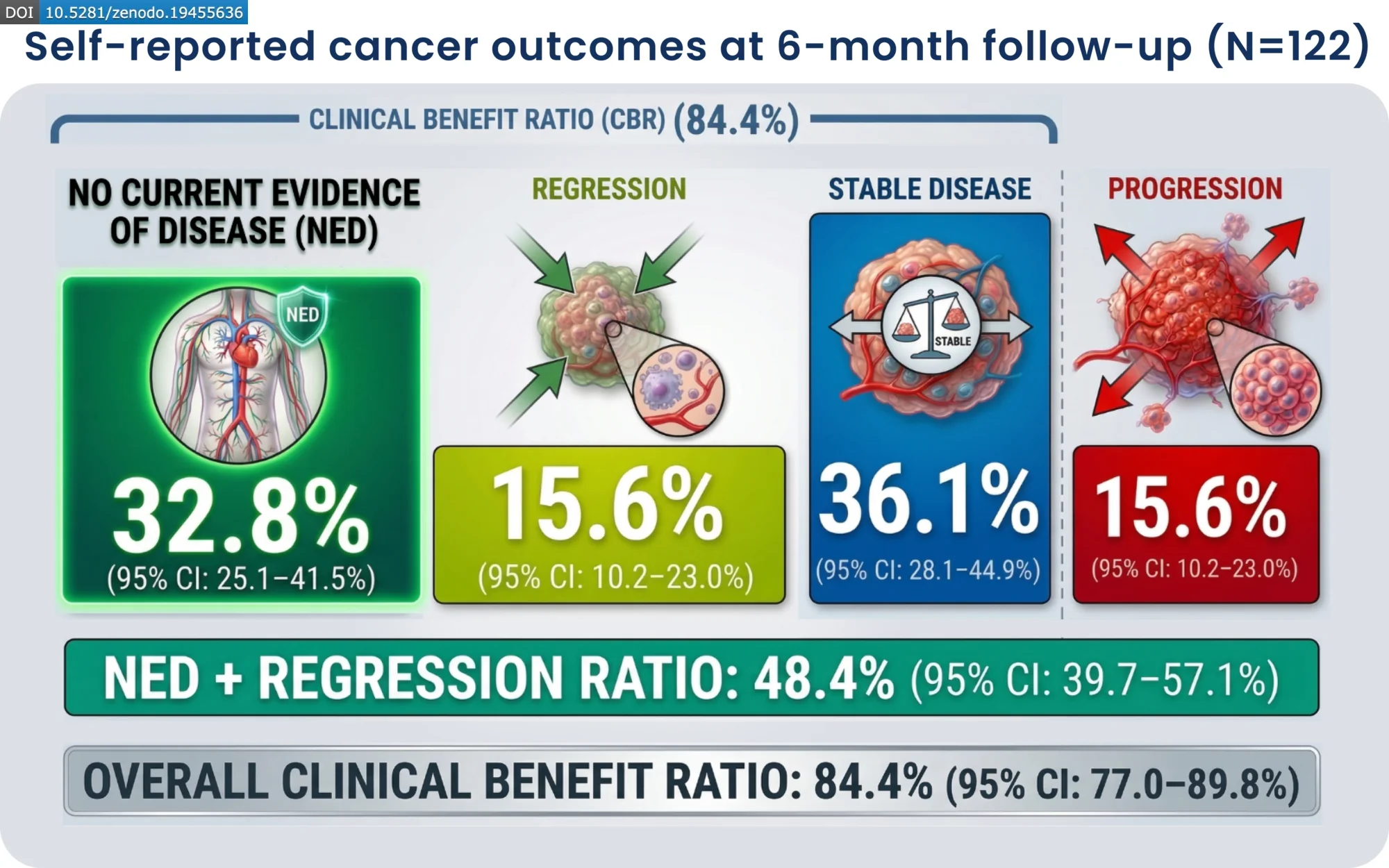
The report analyzed 197 cancer patients who were prescribed ivermectin and mebendazole off-label. Participants received compounded oral capsules containing 25 mg of ivermectin and 250 mg of mebendazole. After 6-months, participants reported an 84% Clinical Benefit Ratio. With 48% of participants reporting regression or no evidence of cancer, while 36% reported disease stability. Only 15.6% of participants reported disease progression.
“In this prospective real-world cohort, the combination of ivermectin and mebendazole was associated with high rates of self-reported clinical benefit, with nearly half of participants reporting tumor regression or no current evidence of disease across a heterogeneous population of cancer patients,” continued McCullough. “These findings provide a compelling clinical signal that these well-tolerated, repurposed agents may offer therapeutic benefit.”
“For far too long, patients have turned to social media ‘experts’ for medical advice,” said Peter Gillooly, CEO of The Wellness Company.
“The American people, particularly when treating devastating illnesses like cancer, deserve answers supported by real-world data, not trending social media posts.”
“This analysis is being done by The Wellness Company for one simple reason,” continued Peter Gillooly. “Cancer patients come to our renowned doctors by the thousands for recommendations on how to handle their diagnosis.”
Cancer remains one of the leading causes of death globally, with conventional treatments such as chemotherapy, radiation therapy, and targeted agents frequently limited by significant toxicity, high cost, development of resistance, and variable long-term efficacy.
In this context, drug repurposing has gained substantial attention as a strategy to rapidly identify effective and affordable therapeutic options using medications with good patient tolerance and well-established safety profiles. This approach offers a practical pathway to accelerate the development of new cancer therapies or accompaniments of therapies while leveraging decades of existing safety data.
According to Dr. Kelly Victory, cancer survivor and co-designer of the analysis, “Prior to this data, ivermectin and mebendazole have demonstrated highly promising anti-cancer activity in preclinical models. But despite compelling preclinical data and documented safe use in cancer patients, robust clinical evidence evaluating the ivermectin–mebendazole combination in oncology remains limited, which is why this analysis was conducted to assess real-world self-reported cancer outcomes, medication adherence, tolerability, and patient experience among individuals prescribed ivermectin and mebendazole.”
“This report reveals an exciting new potential that should expand the consideration of ivermectin and mebendazole for inclusion in the treatment of multiple cancer types,” remarks Dr. Harvey Risch, Professor Emeritus of Epidemiology at Yale School of Public Health and Chair, President’s Cancer Panel at the National Cancer Institute. “We urgently need a full-fledged scientific investigation into this class of medications and their impact on cancer treatment.”
Following the groundbreaking positive results of this report, the authors agree that much more analysis needs to be done, concluding that:
“Given the observational design, reliance on self-reported outcomes, and potential for selection bias and uncontrolled confounding, these findings should be interpreted as hypothesis-generating. Urgent prospective, randomized, placebo-controlled clinical trials are warranted to validate these observations and further define optimal dosing strategies.”
###
Contact Chris Barron at cbarron@rightturnstrategiesdc.com to arrange interviews with Dr. Peter McCullough, Dr. Kelly Victory, Dr. James Thorp, Dr. Drew Pinsky, Dr. Harvey Risch, Nicolas Hulscher or Peter Gillooly.